Home Sleep Tests
How is Sleep Apnea Diagnosed?
The first step in the treatment of sleep apnea is diagnosing the condition and determining the severity of your case. Traditional diagnostic procedures include polysomnography, also called a sleep study, is a test used to diagnose sleep disorders. Polysomnography records your brain waves, the oxygen level in your blood, heart rate, and breathing, as well as eye and leg movements during the study. This is conducted in a sleep laboratory, which involves being connected to uncomfortable equipment and overnight observations.
Sleep Elite often uses one of two home sleep apnea test (HSAT) devices versus sending a patient to a sleep laboratory. This study is a simple, painless procedure that takes place in your home.


WatchPAT®️ ONE
WatchPAT ONE is a revolutionary FDA-Cleared portable diagnostic device that uses innovative technologies to ensure the accurate detection of sleep apnea. Unlike more complicated diagnostic devices, the WatchPAT ONE is easy to use. You wear the watch-like device on your wrist after pairing it with a free app on your iPhone or Android device. You also wear a sensor on your finger.
It was designed with the patient in mind for sleep apnea testing in the comfort of your own bedroom. This environment is more representative of your personal sleep habits. The WatchPAT ONE’s simple operation and robust design ensure the best possible sleep experience.
Within one-minute post-study, the raw data is downloaded and auto-scored identifying all types of apnea events. The simple smartphone app transmits the WatchPAT ONE's data to the cloud. As soon as the study is completed a Sleep Elite clinician can review the automated study results.
It measures up to seven channels:
PAT Signal - Peripheral Arterial Signal
Heart Rate
Oximetry - Measures the oxygen level (oxygen saturation) of the blood.
Actigraphy - A non-invasive method of monitoring human rest/activity cycles.
Body Position
Snoring
Chest Motion
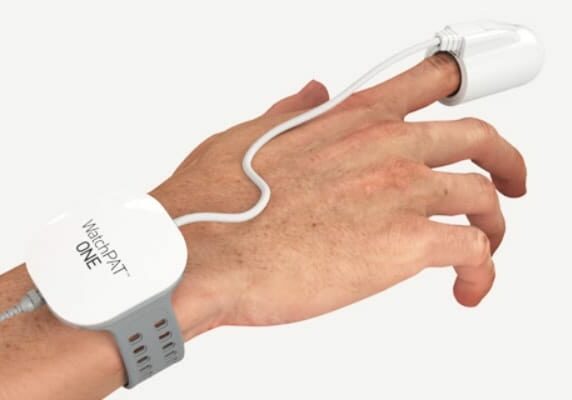


WatchPAT®️ ONE is easy to use
Simply prepare for sleep as you normally do. Just before you go to bed:
- Attach the chest sensor
- Strap on the WatchPAT bracelet to your non-dominate hand
- Slip on the finger probe
That's all there is to it. For more information we can refer you to a step-by-step video.

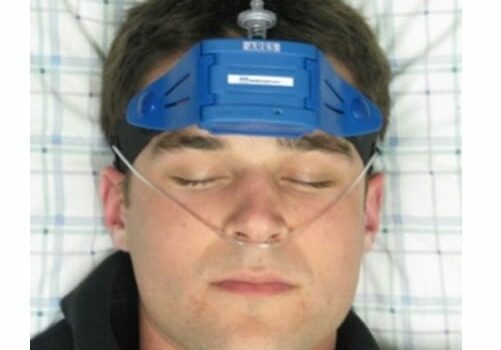
ARES™ Home Sleep Test Device
The Apnea Risk Evaluation System (ARES) integrates physiological data acquired in-home with clinical history and anthropomorphic data to determine the presence and severity of Obstructive Sleep Apnea (OSA). ARES has been cleared for use by the United States Food and Drug Administration (FDA).
A sleep-wearable, wireless physiological recorder worn on the forehead that acquires and stores up to 3 nights of nocturnal data. When worn in the home, the ARES provides a better profile of the patient’s breathing during sleep in his/her normal environment. Audio and visual indicators notify the user when the ARES requires adjustment, thus increasing reliability of the device in the home. The small size of ARES allows it to be comfortably worn in all sleep positions.
ARES measures up to 7 channels:
- Airflow - Nasal pressure
- Pulse Rate
- Snoring
- Head Position/Movement
- Sleep/Wake
- REM/NREM - Rapid Eye Movement and Non-Rapid Eye Movement
- Apnea/Hypopnea Index (AHI)
- Respiratory Disturbance Index (RDI)